Alcohol functional group5/8/2023 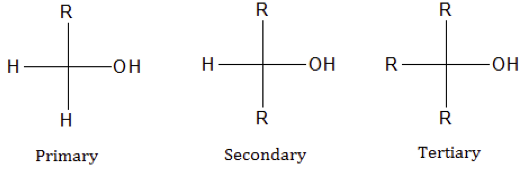
Alcohols have higher boiling and melting points than hydrocarbons with the same number of carbon atoms.Isomers with shorter carbon chains tend to be more soluble because the repulsion between their non-polar regions and water is minimised.įor example, 2-propanol has greater solubility in water than 1-propanol methyl-2-propanol is more soluble than 1-butanol. Polarity and solubility in water also differ between alcohol isomers.Typically, hydrocarbons with more than 7 carbons are considered insoluble in water. For example, methanol and ethanol are very soluble in water whereas octanol is insoluble in water. The solubility of alcohol in water decreases with increasing size (number of carbon atoms).–OH group can donate and accept hydrogen bonds to and from water molecules (shown below). Alcohols are more soluble than hydrocarbons because they can form dipole-dipole forces and hydrogen bonds (due to the presence of hydrogen atom attached to an oxygen atom).Small alcohol molecules tend to be more polar because the polar alcohol functional group contributes more to the overall polarity of the molecule.Larger alcohol molecules tend to be more non-polar because the hydrocarbon region dominates the small alcohol functional group.The overall polarity depends on the size of the non-polar hydrocarbon group. However, the remaining part of an alcohol consists of non-polar hydrocarbons. The alcohol functional group (–OH) is polar due to the high electronegativity of oxygen. 
Alcohol molecules contain polar and non-polar regions.When demonstrating bonding and structure of alcohol, remember to explicitly show the O–H bond (this is often not shown).Structurally, –OH group in carboxylic acids is attached to a sp 2hybridised carbon. Carboxylic acids, despite containing the ‘–OH’ group, are not considered as alcohols as they have very different reactivity.When assigning numbers for their positions along the longest carbon chain, the alcohol functional group is always given the lowest possible number compared to alkenes and alkynes.Alcohol molecules have names ending with the suffix ‘-ol’.In IUPAC nomenclature, the alcohol functional group is assigned higher priority than alkanes, alkenes and alkynes.Methanol (CH 3OH) does not fit in any of the three types of alcohols but it is commonly treated as a primary alcohol because it has a similar reactivity properties.The three types of alcohols share similar properties due to the presence of the same functional group but also differ slightly in their physical as well as chemical properties because the nature of connectivity is different. ‘R’ is a generic representation of an alkyl (carbon) group. There are three types of alcohols (see below). Alcohols are classified as organic molecules which contains a –OH group attached to a sp 3hybridised carbon atom (carbon with only C–C and C–H bonds).Alcohols have different structure, nomenclature and physical properties, such as melting and boiling point and aqueous solubility, when compared to hydrocarbons. This video introduces students to the organic functional group – alcohols. explain the properties within and between the homologous series of alcohols with reference to the intermolecular and intramolecular bonding present – boiling and melting point – solubility in water – molar enthalpy of combustion.investigate the structural formulae, properties and functional group including: – primary – secondary – tertiary alcohols.This is part of the HSC Chemistry course under the topic Alcohols.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
